Mechanical Engineering scholar breakthrough research in cell mechanics by Dr. Lin Yuan
During its development, the embryo must undergo significant axial extension without losing its structural integrity. Failure to execute this process properly may result in fetal abnormality, a grave concern to all prospective parents.
In a recent breakthrough, a research team led by Dr. Yuan Lin has shed critical insight on what causes abnormal embryo elongation, and possible new ways of treating those disorders. Specifically, Dr. Lin and his team showed that active contraction-induced realignment of actin filaments (a major component of cytoskeleton) must be synchronized with the development of intracellular forces for the embryo to elongate, which is then further sustained by muscle contraction-triggered plastic deformation of cells. In addition, it was also found that pre-established anisotropy is essential for the proper onset of the elongation process while defects in the integrity or bundling kinetics of actin filaments result in abnormal embryo extension. The findings have recently been published in Science Advances (https://doi.org/10.1126/sciadv.abg3264).
As a core member in the Biomedical Engineering Programme, Dr. Lin’s lab is among the world’s most active groups in cell mechanics research, particularly in elucidating the physical mechanisms behind important biological processes such as tissue morphogenesis, cell adhesion, cell migration and mechanotransduction, as well as exploring their possible biomedical applications. To achieve these goals, they have been using theoretical modeling and large-scale simulation in conjunction with experimental tools like cutting-edge micro-/nano- fabrication and characterization techniques.
For details, please click here.
Figure 1. Photo of the research team.
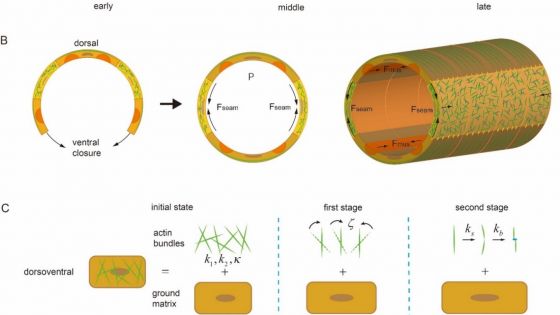
Figure 2. Schematic diagrams of C. elegans embryo elongation. (A) Cartoons showing two stages of elongation driven by seam cell and muscle contractions. (B) Illustration of the microstructure of the embryo wall (treated as a thin-walled cylinder). Contraction generated in seam cells causes the shrinking of the embryo wall in the circumferential direction and eventually drives its axial elongation. Such elongation is further sustained by contraction of body-wall muscles in the second stage. (C) Illustration of the model where the development of cellular anisotropy and plasticity were assumed to be caused by the force-induced alignment and severing/re-bundling of actin filaments, respectively.